Nucleic Acid Insights covers the latest news, trends, issues, and breakthroughs across the rapidly evolving nucleic acids space, including oligonucleotides, mRNA, DNA, formulation and delivery, and more.
Our content - a balance of high-level analysis and in-depth technical data - is designed to give you the understanding and need-to-know information you require to successfully navigate this rapidly evolving space.
We cover critical challenges and the latest innovations in nucleic acid therapies, publishing original research articles, expert reviews, commentary, clinical reports, and more. Visit the Nucleic Acid Insights Journal page for our complete collection.
Our engaging webinar series provides expert-led discussions on significant developments, novel methodologies, and emerging trends, designed to support continued professional growth in nucleic acid research.
Explore our specialised channels, including focused coverage of key areas such as mRNA technology, plasmid DNA production, and oligonucleotide therapeutics, offering targeted insights into manufacturing, delivery, and regulatory considerations critical to therapeutic development.
Learn more about our journal’s mission and publishing criteria by visiting our aims and scope.
Sign-up for free to gain unlimited access to our extensive library of articles, webinars, podcasts, news, and interviews, and stay at the cutting edge of nucleic acid innovation.
If you’re interested in partnering with us through sponsorships, webinars, articles, and more, please view our media kit to learn more.
April 2026
Upcoming webinars
Latest Articles
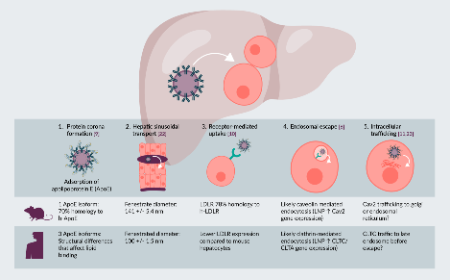
Using humanized liver mice to advance the development of LNP delivery systems

Decoding mRNA stability: from endogenous decay pathways to therapeutic design

Programmable RNA therapeutics: from intracellular sensing to cell-specific delivery

Selective removal of immunogenic double-stranded RNA from mRNA feeds using affinity chromatography

The industry speaks: assessing the state of oligonucleotide manufacturing in 2026

Case studies: Advancing bioprocess analytics with Slope Spectroscopy

Industry Insights: Regulatory progress and AI-driven advances in nucleic acid therapeutics
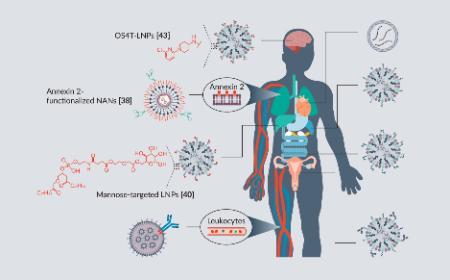
Targeting mRNA therapeutics: current approaches and emerging platforms