Beyond mRNA - how advances in analytical techniques are enabling a revolution of the RNA drug landscape
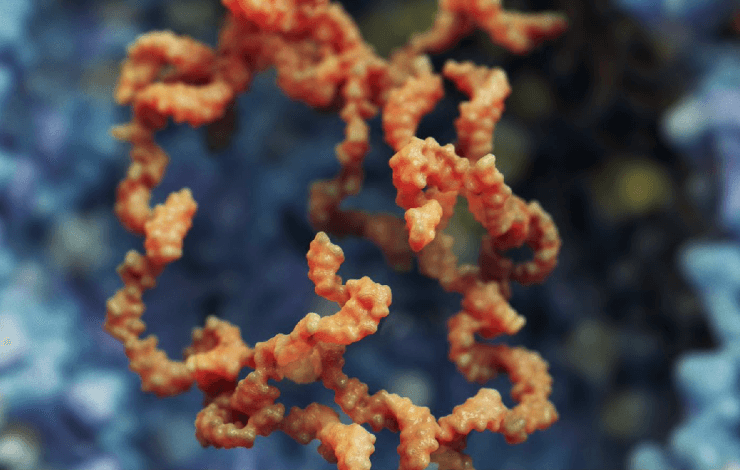
Currently, there are three main types of in vitro transcribed (IVT) RNA drugs: conventional, non-replicating mRNA; base-modified, non-replicating mRNA (bmRNA), which incorporate chemically modified nucleotides; and self-replicating RNA (srRNA), based on an engineered viral genome but devoid of viral structural protein genes.
The ability to create copies of the drug after administration enables lower dosing and expands the range of potential indications. However, srRNAs are often much larger in size (9,000-16,000 bases) and traditional biophysical analytical techniques, such as capillary electrophoresis (CE), have been challenging to adapt to these large molecules.
This presentation will showcase how to achieve improved characterization of srRNA using the newest advances in CE. Additionally, we will discuss how these complement and augment in vitro cell-based assays currently being used for potency testing.
- Learn about the potential of srRNA as a vaccine and therapeutic modality
- Explore how advances in CE can overcome analytical difficulties when assessing RNA up to 16,000 bases
- Hear about how cell-based potency assays correlate with purity and size assessment with capillary gel electrophoresis
You might also like

Assessing mRNA stability and integrity using mass photometry
Exploring the latest advances in purification and analysis of oligonucleotide therapeutics
A scalable purification strategy for removal of dsRNA byproducts following IVT RNA production

Tools, trends, and technologies: analytical development in ADCs


