RNA interference therapies targeting peritoneal macrophages for local and systemic diseases
Nucleic Acid Insights 2026; 3(2), 109–131
DOI: 10.18609/nuc.2026.014
The peritoneal cavity serves as a unique immunological niche where peritoneal macrophages (PMs) play central roles in local and systemic immunity. Their abundance, accessibility, and phenotypic plasticity make them attractive targets for RNA interference (RNAi) based therapies. By silencing disease-driving gene expression via RNAi effectors, PMs can be directed towards pro- or anti-inflammatory phenotypes. While direct intraperitoneal (IP) delivery of short-interfering RNA (siRNA) offers access to PMs, variable distribution and rapid clearance reduce efficiency and underscore the need for optimized delivery strategies. Nanoparticle carriers are employed to improve the distribution and delivery efficiency of RNAi effectors and are capable of enhancing uptake into PMs. Ligand-targeting through macrophage-specific receptors further promotes specificity and cellular internalization. Collectively, these strategies utilizing RNAi technology to target large peritoneal macrophages (LPMs) represent promising approaches for the treatment of local diseases such as peritoneal cancer, endometriosis, peritonitis, and systemic diseases such as sepsis, autoimmunity, and atherosclerosis. Key challenges in therapeutic development include inefficient endosomal escape, excessive off-target uptake, and unwanted immune activation. Addressing these challenges is essential for advancing macrophage‑targeted therapies toward successful clinical translation.
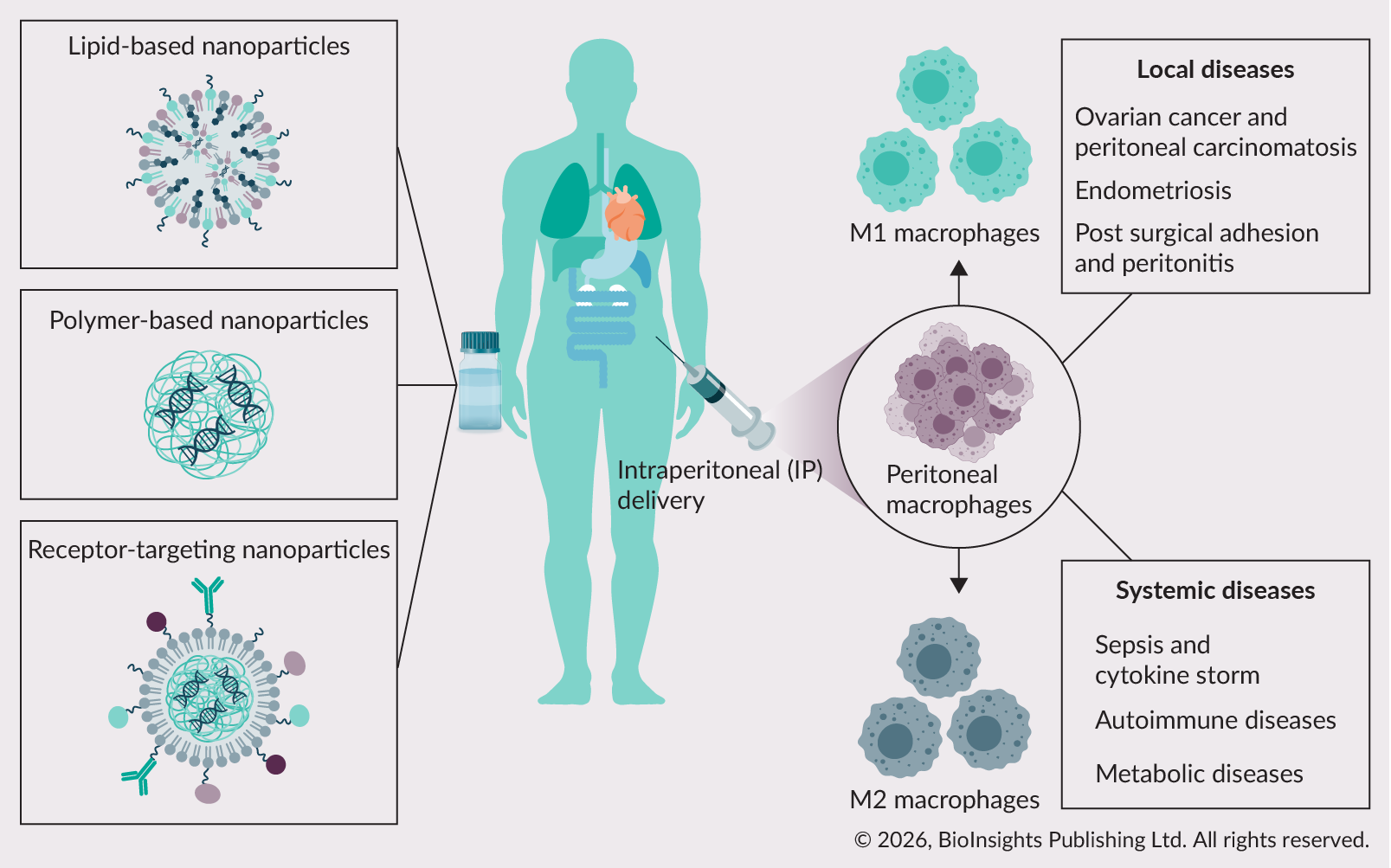 |
| Graphical abstract. © 2026, BioInsights Publishing Ltd. All rights reserved. |
