New analytical method for rAAV titer and empty/full ratio determination in cell lysate and conditioned medium
DOI: 10.18609/cgti.2026.030
Cell & Gene Therapy Insights 2026; 12(2), 241–249
Recombinant AAV (rAAV) is widely used as a gene delivery vector for in vivo gene therapy; however empty particles (EPs) remain a major challenge. Two main strategies can be considered to reduce EPs while maintaining sufficient titer: (1) increasing the full particle (FP) ratio in total particles (F%) through upstream process optimization, and (2) enriching FPs through downstream processes such as chromatography. When pursuing an upstream approach, it is desirable to systematically vary transfection parameters and culture conditions to identify optimal conditions by evaluating both titer and F% in crude cell lysate and conditioned medium. In this context, analytical methods that can quantify titer and F% in a simple yet highly reliable manner are essential.
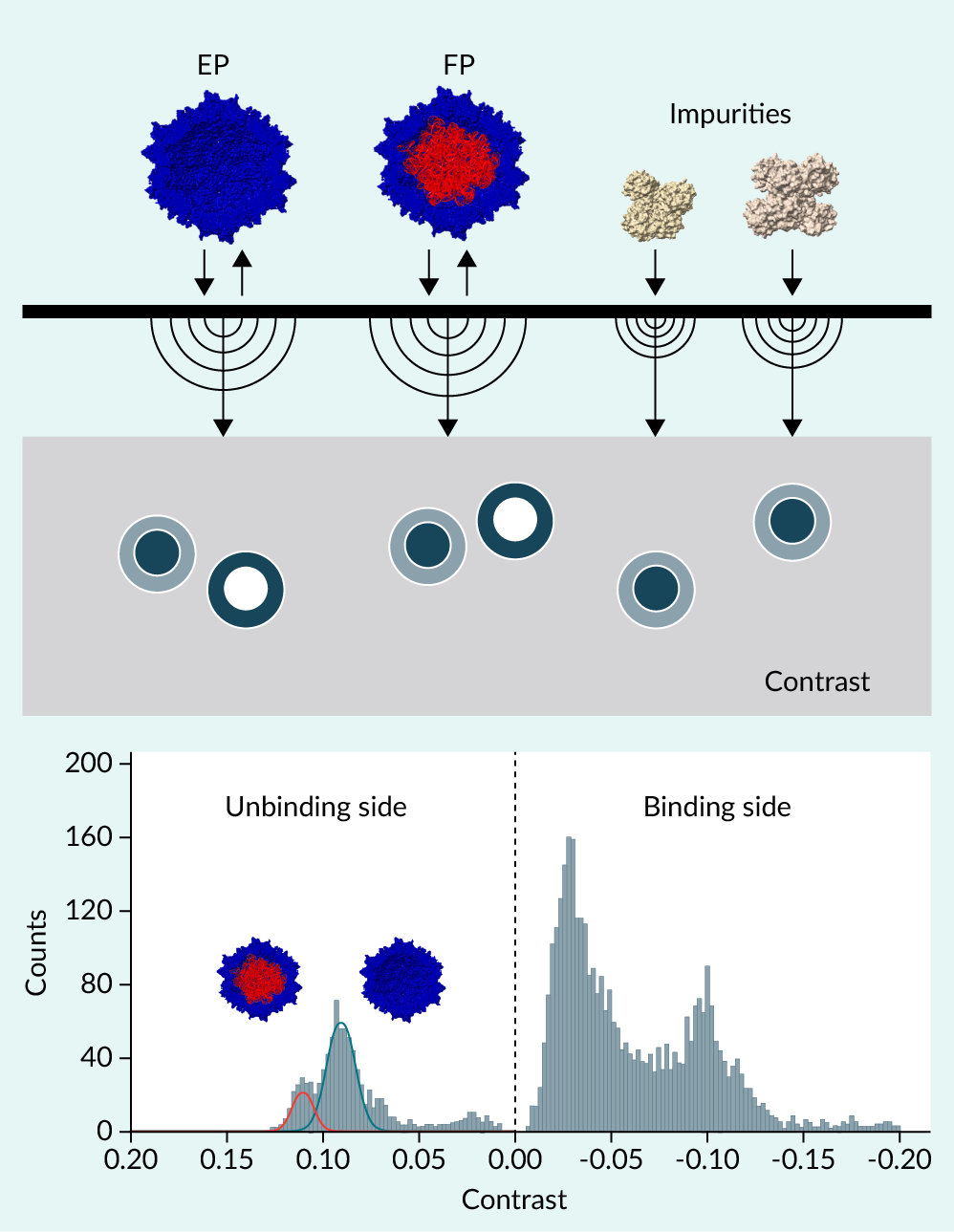
In this article, we present the ‘DirectMP method’, which we recently developed for direct evaluation of both titer and F% in crude cell lysate and conditioned medium without the need for special sample pretreatment. Originally established on the TwoMP platform, DirectMP also showed potential applicability to SamuxMP, as demonstrated using lysates of cells producing AAV serotype 8. A key feature of DirectMP is its analysis of signals from molecules unbinding from the glass surface, whereas conventional mass photometry (MP) relies on signals from molecules binding to the glass surface. By using unbinding signals, DirectMP suppresses interference from impurities, enabling extraction and analysis of rAAV-derived signals while retaining the key advantages of MP, namely rapid mass measurement with minimal sample consumption. This method is expected to be applicable to a wide range of rAAV serotypes and to contribute significantly to upstream process optimization and overall improvement of rAAV manufacturing.
rAAV production yields both full particles (FPs) and empty particles (EPs). High EP burden increases immunogenic load and complicates upstream optimisation.
ELISA/PCR – limited F% reliability.
Dual FLISA – 4–5 h, needs standards.